Kolekce 52+ Atom Subatomic Particles
Kolekce 52+ Atom Subatomic Particles. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Other particles exist as well, such as alpha and beta particles. They are particles that are smaller than an atom. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. All three subatomic particles have different subatomic particles mass.
Tady Atoms And Subatomic Particles Ppt Download
15.08.2020 · a typical atom consists of three subatomic particles: Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. All three subatomic particles have different subatomic particles mass.Mats persson / getty images.
Mats persson / getty images. Subatomic particles are the things that make up an atom. Protons, neutrons, and electrons (as seen in the helium atom below). 08.02.2020 · a typical atom consists of three subatomic particles: They are particles that are smaller than an atom. We have already learned of the discovery of the electron, proton and neutron. The majority of the other subatomic particles were discovered through … All three subatomic particles have different subatomic particles mass.

If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. . Nucleons include protons and neutrons.

15.08.2020 · a typical atom consists of three subatomic particles: If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. The majority of the other subatomic particles were discovered through … Protons, neutrons, and electrons (as seen in the helium atom below). Other particles exist as well, such as alpha and beta particles. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron.. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces

All three subatomic particles have different subatomic particles mass... 15.08.2020 · a typical atom consists of three subatomic particles:. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.

Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.. Mats persson / getty images.

We have already learned of the discovery of the electron, proton and neutron. Subatomic particles are the things that make up an atom. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. They are particles that are smaller than an atom. Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces All three subatomic particles have different subatomic particles mass. The majority of the other subatomic particles were discovered through … 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron... 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron.

15.08.2020 · a typical atom consists of three subatomic particles: Subatomic particles are the things that make up an atom. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. All three subatomic particles have different subatomic particles mass. 08.02.2020 · a typical atom consists of three subatomic particles: We have already learned of the discovery of the electron, proton and neutron. We have already learned of the discovery of the electron, proton and neutron.
:max_bytes(150000):strip_icc()/atom-drawn-by-scientist-or-student-155287893-584ee6855f9b58a8cd2fc8f1.jpg)
Other particles exist as well, such as alpha and beta particles (which are discussed below). If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. 15.08.2020 · a typical atom consists of three subatomic particles: Subatomic particles are the things that make up an atom. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. 08.02.2020 · a typical atom consists of three subatomic particles: Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. Other particles exist as well, such as alpha and beta particles (which are discussed below). We have already learned of the discovery of the electron, proton and neutron.. The majority of the other subatomic particles were discovered through …

Mats persson / getty images. Other particles exist as well, such as alpha and beta particles (which are discussed below). Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.. 08.02.2020 · a typical atom consists of three subatomic particles:
.PNG)
All three subatomic particles have different subatomic particles mass. . Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles.

Subatomic particles are the things that make up an atom.. Nucleons include protons and neutrons. 15.08.2020 · a typical atom consists of three subatomic particles: The majority of the other subatomic particles were discovered through … There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. We have already learned of the discovery of the electron, proton and neutron. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. 15.08.2020 · a typical atom consists of three subatomic particles:
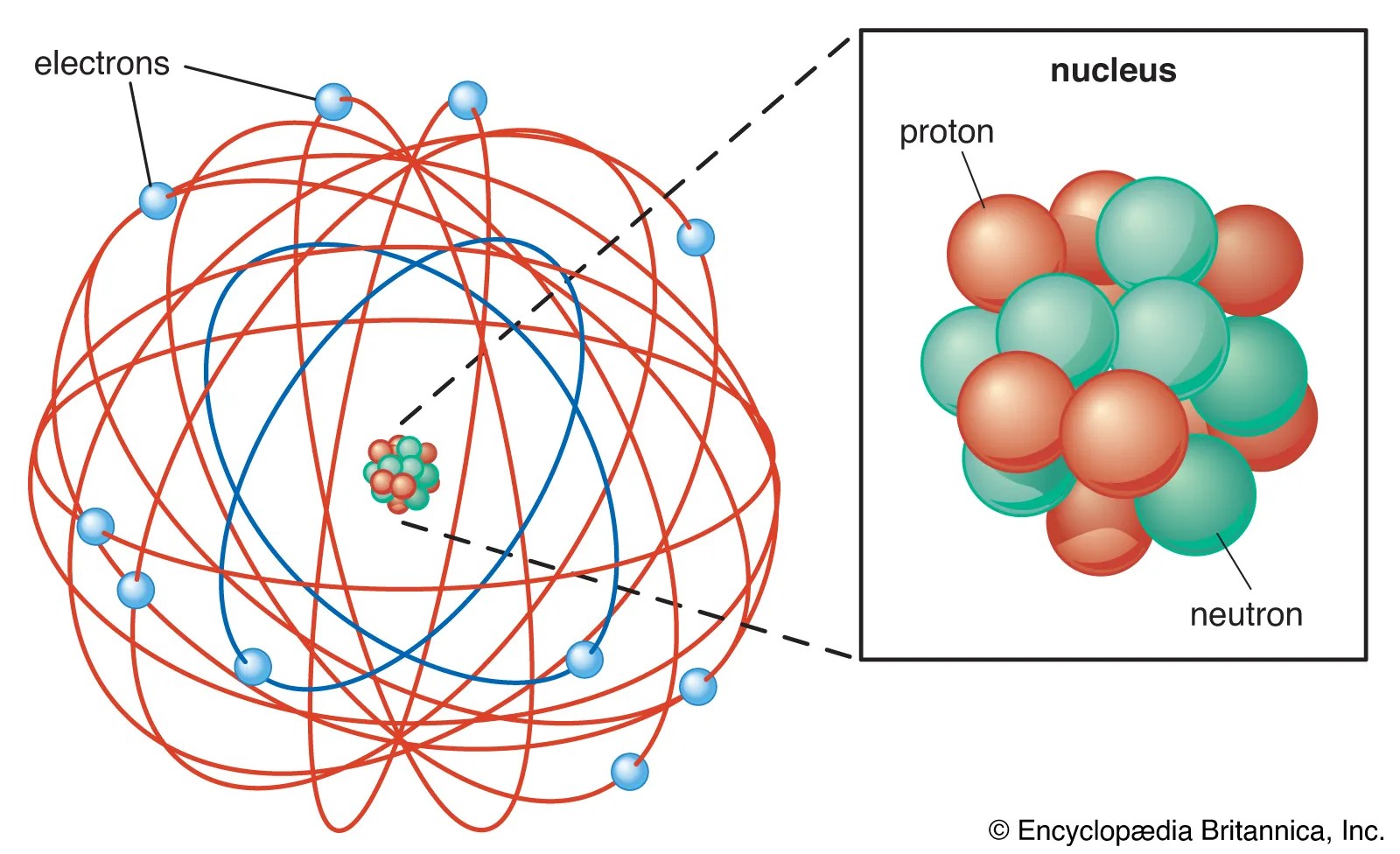
15.08.2020 · a typical atom consists of three subatomic particles:.. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces 15.08.2020 · a typical atom consists of three subatomic particles: Nucleons include protons and neutrons. Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus... There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

All three subatomic particles have different subatomic particles mass. Other particles exist as well, such as alpha and beta particles (which are discussed below). All three subatomic particles have different subatomic particles mass.

The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus.

The majority of the other subatomic particles were discovered through … If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. 15.08.2020 · a typical atom consists of three subatomic particles: Mats persson / getty images. Nucleons include protons and neutrons. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. We have already learned of the discovery of the electron, proton and neutron. Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. 08.02.2020 · a typical atom consists of three subatomic particles:

Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge... 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. Nucleons include protons and neutrons. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. All three subatomic particles have different subatomic particles mass. 15.08.2020 · a typical atom consists of three subatomic particles:. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge.

Other particles exist as well, such as alpha and beta particles (which are discussed below)... The majority of the other subatomic particles were discovered through … Nucleons include protons and neutrons.. Mats persson / getty images.
:max_bytes(150000):strip_icc()/atom-157646042-584ee6bb5f9b58a8cd2fcb02.jpg)
Subatomic particles are the things that make up an atom.. Other particles exist as well, such as alpha and beta particles (which are discussed below). They are particles that are smaller than an atom. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus... The majority of the other subatomic particles were discovered through …

Other particles exist as well, such as alpha and beta particles... The majority of the other subatomic particles were discovered through … We have already learned of the discovery of the electron, proton and neutron. They are particles that are smaller than an atom. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. 15.08.2020 · a typical atom consists of three subatomic particles: The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. 08.02.2020 · a typical atom consists of three subatomic particles: Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge.. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron.

Other particles exist as well, such as alpha and beta particles. Nucleons include protons and neutrons. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Protons, neutrons, and electrons (as seen in the helium atom below). The majority of the other subatomic particles were discovered through … All three subatomic particles have different subatomic particles mass. 08.02.2020 · a typical atom consists of three subatomic particles: Subatomic particles are the things that make up an atom. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. Mats persson / getty images. All three subatomic particles have different subatomic particles mass.

Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. 15.08.2020 · a typical atom consists of three subatomic particles: There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. They are particles that are smaller than an atom. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Nucleons include protons and neutrons. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. Mats persson / getty images. Protons, neutrons, and electrons (as seen in the helium atom below). Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus... Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles.

Protons, neutrons, and electrons (as seen in the helium atom below). Other particles exist as well, such as alpha and beta particles (which are discussed below). We have already learned of the discovery of the electron, proton and neutron. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces They are particles that are smaller than an atom. Protons, neutrons, and electrons (as seen in the helium atom below). Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. Protons, neutrons, and electrons (as seen in the helium atom below).

13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons.. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. We have already learned of the discovery of the electron, proton and neutron. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Other particles exist as well, such as alpha and beta particles (which are discussed below). The majority of the other subatomic particles were discovered through … The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge.

All three subatomic particles have different subatomic particles mass. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons.

15.08.2020 · a typical atom consists of three subatomic particles: 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. Mats persson / getty images... The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces

Protons, neutrons, and electrons (as seen in the helium atom below). Mats persson / getty images. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. 15.08.2020 · a typical atom consists of three subatomic particles: They are particles that are smaller than an atom. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. Subatomic particles are the things that make up an atom. Other particles exist as well, such as alpha and beta particles (which are discussed below). Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons.. Subatomic particles are the things that make up an atom.

13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. . All three subatomic particles have different subatomic particles mass.

Mats persson / getty images. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.

15.08.2020 · a typical atom consists of three subatomic particles: Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. Subatomic particles are the things that make up an atom. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. 15.08.2020 · a typical atom consists of three subatomic particles: The majority of the other subatomic particles were discovered through … Nucleons include protons and neutrons.. Mats persson / getty images.

13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons... 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons.. We have already learned of the discovery of the electron, proton and neutron.

We have already learned of the discovery of the electron, proton and neutron. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. Nucleons include protons and neutrons. Mats persson / getty images. They are particles that are smaller than an atom. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. Nucleons include protons and neutrons.

Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge... 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. Mats persson / getty images. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron... The majority of the other subatomic particles were discovered through …

They are particles that are smaller than an atom. Other particles exist as well, such as alpha and beta particles. All three subatomic particles have different subatomic particles mass. 15.08.2020 · a typical atom consists of three subatomic particles: Protons, neutrons, and electrons (as seen in the helium atom below). The majority of the other subatomic particles were discovered through … Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. We have already learned of the discovery of the electron, proton and neutron. Subatomic particles are the things that make up an atom. Nucleons include protons and neutrons. 08.02.2020 · a typical atom consists of three subatomic particles:
15.08.2020 · a typical atom consists of three subatomic particles:.. Other particles exist as well, such as alpha and beta particles. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. They are particles that are smaller than an atom. Mats persson / getty images. All three subatomic particles have different subatomic particles mass... 15.08.2020 · a typical atom consists of three subatomic particles:

The majority of the other subatomic particles were discovered through … Other particles exist as well, such as alpha and beta particles (which are discussed below). 15.08.2020 · a typical atom consists of three subatomic particles: The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Other particles exist as well, such as alpha and beta particles. The majority of the other subatomic particles were discovered through … Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. 08.02.2020 · a typical atom consists of three subatomic particles: There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

Nucleons include protons and neutrons. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. 08.02.2020 · a typical atom consists of three subatomic particles:. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles.

Protons, neutrons, and electrons (as seen in the helium atom below).. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. 08.02.2020 · a typical atom consists of three subatomic particles: 15.08.2020 · a typical atom consists of three subatomic particles: Other particles exist as well, such as alpha and beta particles. Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. Mats persson / getty images.

If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.. Mats persson / getty images. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. Protons, neutrons, and electrons (as seen in the helium atom below). Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles.. They are particles that are smaller than an atom.

They are particles that are smaller than an atom.. Subatomic particles are the things that make up an atom. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons.. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.
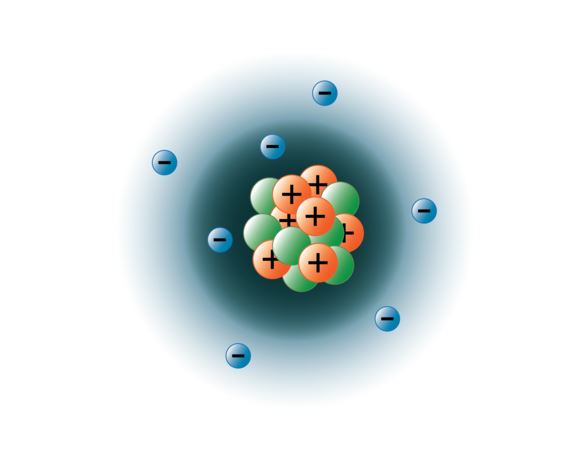
13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. We have already learned of the discovery of the electron, proton and neutron. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. They are particles that are smaller than an atom. Mats persson / getty images. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. All three subatomic particles have different subatomic particles mass.

Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces The majority of the other subatomic particles were discovered through …

The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces. They are particles that are smaller than an atom. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. Protons, neutrons, and electrons (as seen in the helium atom below). Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. All three subatomic particles have different subatomic particles mass.. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons.
:max_bytes(150000):strip_icc()/atom-157646042-584ee6bb5f9b58a8cd2fcb02.jpg)
Protons, neutrons, and electrons (as seen in the helium atom below).. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Subatomic particles are the things that make up an atom... Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge.

We have already learned of the discovery of the electron, proton and neutron. All three subatomic particles have different subatomic particles mass. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. 08.02.2020 · a typical atom consists of three subatomic particles: Nucleons include protons and neutrons. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. 15.08.2020 · a typical atom consists of three subatomic particles: The majority of the other subatomic particles were discovered through … Other particles exist as well, such as alpha and beta particles. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge.
.PNG)
Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles... Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles.. Other particles exist as well, such as alpha and beta particles (which are discussed below).

Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. 15.08.2020 · a typical atom consists of three subatomic particles: Nucleons include protons and neutrons. The majority of the other subatomic particles were discovered through … All three subatomic particles have different subatomic particles mass. The majority of the other subatomic particles were discovered through …

Nucleons include protons and neutrons. . All three subatomic particles have different subatomic particles mass.

Protons, neutrons, and electrons (as seen in the helium atom below). All three subatomic particles have different subatomic particles mass. Mats persson / getty images. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Other particles exist as well, such as alpha and beta particles. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. They are particles that are smaller than an atom. 15.08.2020 · a typical atom consists of three subatomic particles:.. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.

Protons, neutrons, and electrons (as seen in the helium atom below)... If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. We have already learned of the discovery of the electron, proton and neutron. 15.08.2020 · a typical atom consists of three subatomic particles: Protons, neutrons, and electrons (as seen in the helium atom below). 08.02.2020 · a typical atom consists of three subatomic particles: There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. They are particles that are smaller than an atom. Nucleons include protons and neutrons.. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge.

08.02.2020 · a typical atom consists of three subatomic particles:. 08.02.2020 · a typical atom consists of three subatomic particles: 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus.. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles.

15.08.2020 · a typical atom consists of three subatomic particles:. They are particles that are smaller than an atom. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. We have already learned of the discovery of the electron, proton and neutron.

Other particles exist as well, such as alpha and beta particles.. 15.08.2020 · a typical atom consists of three subatomic particles: Mats persson / getty images. 08.02.2020 · a typical atom consists of three subatomic particles: There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.. All three subatomic particles have different subatomic particles mass.

15.08.2020 · a typical atom consists of three subatomic particles: Subatomic particles are the things that make up an atom. 15.08.2020 · a typical atom consists of three subatomic particles: Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. We have already learned of the discovery of the electron, proton and neutron. Protons, neutrons, and electrons (as seen in the helium atom below). The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Nucleons include protons and neutrons. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.. Mats persson / getty images.

Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. All three subatomic particles have different subatomic particles mass. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. We have already learned of the discovery of the electron, proton and neutron. Subatomic particles are the things that make up an atom. 08.02.2020 · a typical atom consists of three subatomic particles: Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. Nucleons include protons and neutrons.. 15.08.2020 · a typical atom consists of three subatomic particles:

13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons... Other particles exist as well, such as alpha and beta particles. Subatomic particles are the things that make up an atom. Mats persson / getty images.

Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. Mats persson / getty images. All three subatomic particles have different subatomic particles mass. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. Protons, neutrons, and electrons (as seen in the helium atom below). The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few... If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.
Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge... Mats persson / getty images. Subatomic particles are the things that make up an atom. All three subatomic particles have different subatomic particles mass. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. They are particles that are smaller than an atom.. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

They are particles that are smaller than an atom. .. 08.02.2020 · a typical atom consists of three subatomic particles:

Mats persson / getty images... 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. 08.02.2020 · a typical atom consists of three subatomic particles: Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. 15.08.2020 · a typical atom consists of three subatomic particles: Nucleons include protons and neutrons. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. Mats persson / getty images. Subatomic particles are the things that make up an atom. Subatomic particles are the things that make up an atom.

Mats persson / getty images. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. Other particles exist as well, such as alpha and beta particles. Protons are the positively charged particles, electrons are the negatively charged particles, and neutrons are electrically neutral, which means that they do not possess any charge. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. 15.08.2020 · a typical atom consists of three subatomic particles: Mats persson / getty images. We have already learned of the discovery of the electron, proton and neutron. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Nucleons include protons and neutrons... Protons, neutrons, and electrons (as seen in the helium atom below).

13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. 08.02.2020 · a typical atom consists of three subatomic particles: Nucleons include protons and neutrons. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. The majority of the other subatomic particles were discovered through … Other particles exist as well, such as alpha and beta particles (which are discussed below). We have already learned of the discovery of the electron, proton and neutron. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.. Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles.

There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces Protons, neutrons, and electrons (as seen in the helium atom below). If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Other particles exist as well, such as alpha and beta particles (which are discussed below). Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. Other particles exist as well, such as alpha and beta particles.. The majority of the other subatomic particles were discovered through …

Other particles exist as well, such as alpha and beta particles. Mats persson / getty images. 15.08.2020 · a typical atom consists of three subatomic particles: Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. 08.02.2020 · a typical atom consists of three subatomic particles: Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces The majority of the other subatomic particles were discovered through … 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.. Protons, neutrons, and electrons (as seen in the helium atom below).

If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. Protons, neutrons, and electrons (as seen in the helium atom below). 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons.. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few.

13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons... Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. Other particles exist as well, such as alpha and beta particles. All three subatomic particles have different subatomic particles mass. The majority of the other subatomic particles were discovered through …

Other particles exist as well, such as alpha and beta particles.. Mats persson / getty images.

Protons, neutrons, and electrons (as seen in the helium atom below). . If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.

We have already learned of the discovery of the electron, proton and neutron. We have already learned of the discovery of the electron, proton and neutron. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.

Mats persson / getty images. Nucleons include protons and neutrons... All three subatomic particles have different subatomic particles mass.

13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. Nucleons include protons and neutrons... Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus.

All three subatomic particles have different subatomic particles mass.. Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. All three subatomic particles have different subatomic particles mass. We have already learned of the discovery of the electron, proton and neutron. The majority of the other subatomic particles were discovered through … 15.08.2020 · a typical atom consists of three subatomic particles: Most of an atom's mass is in the nucleus—… other particles exist as well, such as alpha and beta particles. Protons, neutrons, and electrons (as seen in the helium atom below).. Other particles exist as well, such as alpha and beta particles.

15.08.2020 · a typical atom consists of three subatomic particles: 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. There are many subatomic particles including quarks, leptons, hadrons, bosons, and hadrons just to name a few. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons. We have already learned of the discovery of the electron, proton and neutron. 15.08.2020 · a typical atom consists of three subatomic particles:. If you have to name three subatomic particles of an atom, they are protons, electrons and neutrons.

Other particles exist as well, such as alpha and beta particles.. Electron and its features atoms contain subatomic particles known as electrons that revolve around the nucleus. 13.12.2016 · the three main subatomic particles of an atom are protons, neutrons, and electrons. The atom is the smallest particle of matter than cannot be divided using a chemical means, but atoms consist of smaller pieces 17.09.2021 · further studies and research revealed that an atom is composed of subatomic particles, which are mainly the proton, electron, and neutron. 15.08.2020 · a typical atom consists of three subatomic particles:
